Kinetic Theory of Gases - Complete Guide for JEE 2025-26
Why Kinetic Theory of Gases is Important for JEE?
Kinetic Theory of Gases is a high-scoring chapter in JEE Physics that bridges microscopic molecular behavior with macroscopic gas properties. Understanding this chapter is crucial for:
- Thermodynamics - Foundation for understanding heat and work
- Statistical Mechanics - Links to advanced physics concepts
- Chemical Kinetics - Explains reaction rates and activation energy
- Real Gas Behavior - Understanding deviations from ideal behavior
In JEE Main, expect 2-3 direct questions (8-12 marks). In JEE Advanced, this chapter often appears in multi-concept problems worth 10-16 marks.
Key Topics & Must-Remember Formulas
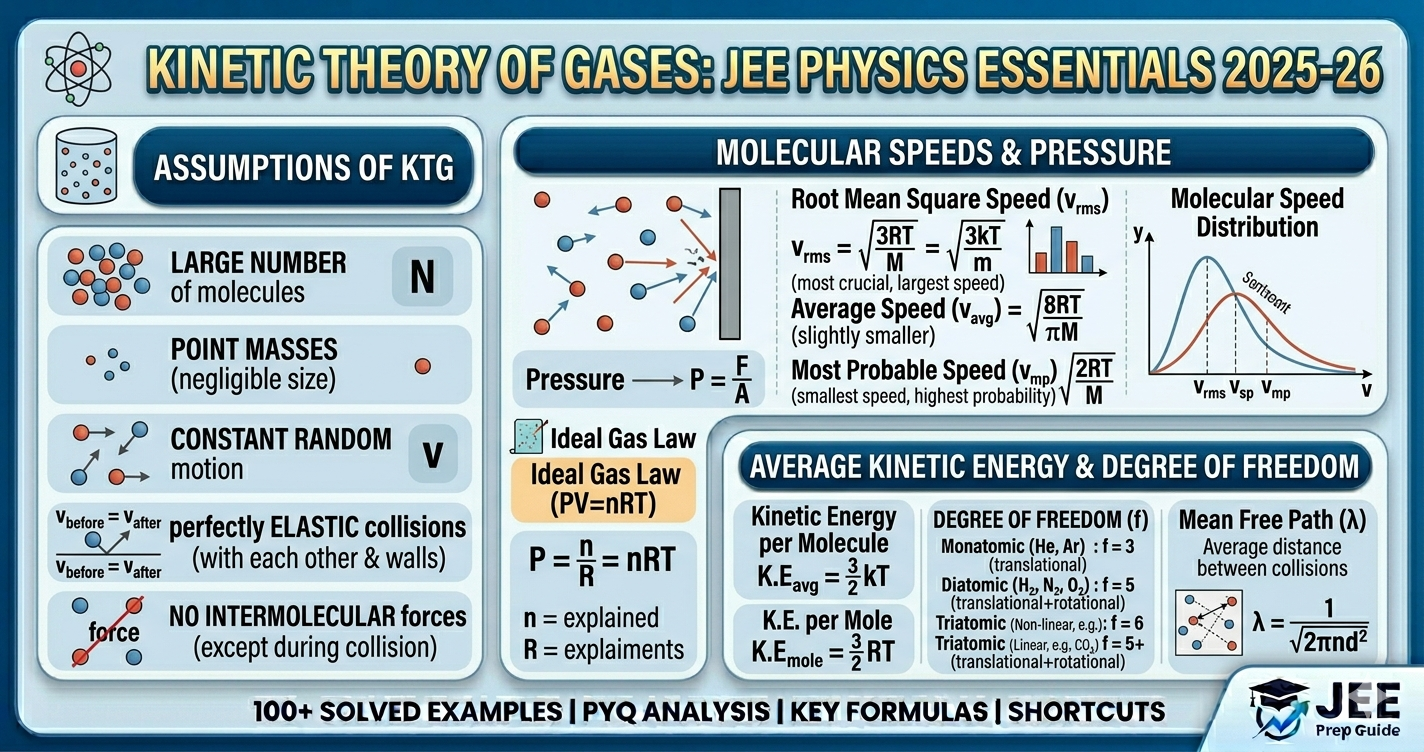
1. Three Molecular Speeds
v_mp : v_avg : v_rms = √2 : √(8/π) : √3 = 1 : 1.128 : 1.224
2. Ideal Gas Equation Forms
- • PV = nRT (standard)
- • PV = NkT (molecular form)
- • P = (ρ/M)RT (density form)
- • P = (1/3)ρv²_rms (kinetic theory)
3. Degrees of Freedom
Monoatomic: f=3, γ=5/3 | Diatomic: f=5, γ=7/5 | Polyatomic: f=6, γ=4/3
4. Mean Free Path
λ = 1/(√2πd²n) = kT/(√2πd²P)
📚 How to Master Kinetic Theory of Gases?
For JEE Main Students:
- Time Required: 4-5 days (3 hours/day)
- Master all three molecular speed formulas and their ratios
- Practice 60+ numerical problems on gas laws and ideal gas equation
- Memorize γ, Cv, Cp values for all three types of gases
- Understand mean free path concept and solve at least 15 problems
- Focus on numerical-heavy topics (speeds, energies, pressures)
For JEE Advanced Students:
- Time Required: 6-7 days (4 hours/day)
- Master all derivations (pressure, mean free path, distribution)
- Practice multi-concept problems combining with thermodynamics
- Deep understanding of Maxwell-Boltzmann distribution
- Solve assertion-reason and multi-correct type questions
- Study applications: Graham's law, diffusion, effusion
⚠️ Common Mistakes to Avoid in JEE Exam
-
❌
Using wrong speed formula: Use v_rms for pressure calculations, v_avg for collision rates, v_mp for Maxwell distribution peak
-
❌
Forgetting to convert temperature to Kelvin: All gas law calculations require absolute temperature (K), not Celsius
-
❌
Wrong γ values: Monoatomic γ=5/3, Diatomic γ=7/5, Polyatomic γ=4/3 (memorize this!)
-
❌
Molecular mass units: Use M in kg/mol for SI units, not g/mol (multiply by 10⁻³)
-
❌
Mean free path confusion: λ ∝ 1/P (inversely proportional to pressure at constant T)
-
❌
Equipartition theorem: Each degree of freedom contributes (1/2)kT per molecule, NOT per mole
📊 JEE Previous Year Question Analysis (2015-2024)
| Year | JEE Main | JEE Advanced | Topic Focus |
|---|---|---|---|
| 2024 | 2 Questions (8 marks) | 3 Questions (12 marks) | Molecular speeds, Mean free path |
| 2023 | 3 Questions (12 marks) | 2 Questions (10 marks) | Degrees of freedom, Specific heats |
| 2022 | 2 Questions (8 marks) | 4 Questions (15 marks) | Gas mixtures, Maxwell distribution |
| 2021 | 2 Questions (8 marks) | 3 Questions (11 marks) | Ideal gas equation, Kinetic energy |
Trend: JEE Advanced increasingly focuses on conceptual understanding and multi-step problems. Pure formula-based questions are becoming rare. Integration with thermodynamics is a common pattern.
🎯 Quick Formula Sheet (Must Memorize)
Molecular Speeds
v_rms = √(3RT/M) = √(3P/ρ)
v_avg = √(8RT/πM)
v_mp = √(2RT/M)
Energy Relations
KE per molecule = (3/2)kT
KE per mole = (3/2)RT
Internal Energy U = (f/2)nRT
Specific Heats
Cv = (f/2)R
Cp = Cv + R = [(f+2)/2]R
γ = Cp/Cv = (f+2)/f
Mean Free Path
λ = 1/(√2πd²n)
λ = kT/(√2πd²P)
Collision frequency ν = v_avg/λ