Thermal Properties of Matter - Complete Guide for JEE 2025-26
Why Thermal Properties is Crucial for JEE?
Thermal Properties of Matter is a high-scoring chapter in JEE Physics with consistent 8-12% weightage. The numerical problems are formula-based and scoring if you practice well.
- Calorimetry - Guaranteed 1-2 questions (mixing, phase change)
- Thermal Expansion - Railway track, pendulum clock problems
- Heat Transfer - Conduction (compound bar) most important
- Newton's Cooling - High probability in JEE Advanced
- Radiation - Stefan's law, Wien's law applications
In JEE Advanced, expect multi-concept questions combining calorimetry with thermodynamics or kinetic theory.
Key Formulas & Values to Memorize
1. Must-Remember Constants
- • c_water = 4200 J/kg·K = 1 cal/g·°C
- • L_f (ice) = 334 kJ/kg = 80 cal/g
- • L_v (water) = 2260 kJ/kg = 540 cal/g
- • σ (Stefan) = 5.67 × 10⁻⁸ W/m²·K⁴
- • b (Wien) = 2.9 × 10⁻³ m·K
2. Key Formulas
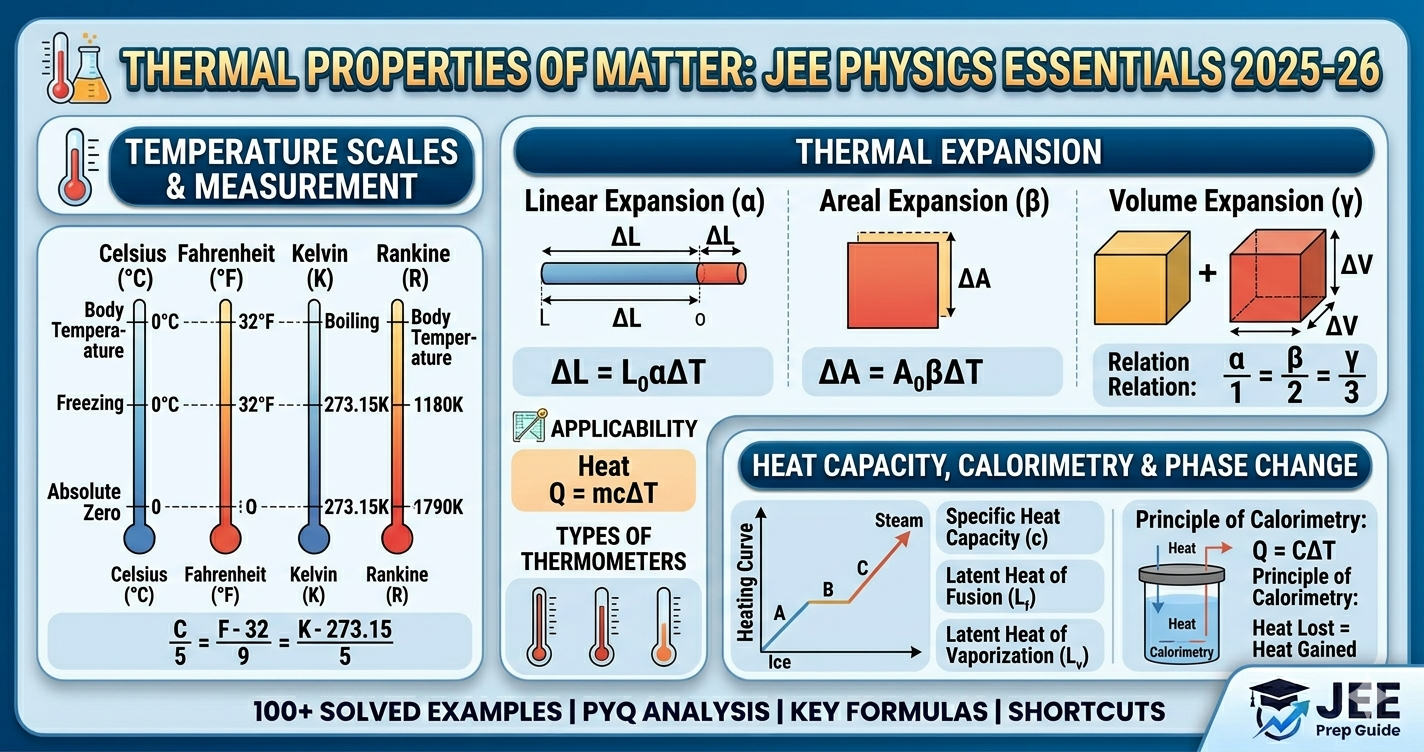
- • Heat: Q = mcΔT
- • Latent heat: Q = mL
- • Linear expansion: ΔL = αL₀ΔT (β=2α, γ=3α)
- • Conduction: dQ/dt = kA(T₁-T₂)/L
- • Radiation: P = eσAT⁴
- • Newton's cooling: dT/dt = -k(T-T₀)
3. Special Relations
Thermal resistance: R = L/(kA) | Series: R_eq = R₁+R₂ | Parallel: 1/R_eq = 1/R₁+1/R₂
📚 How to Master Thermal Properties for JEE?
For JEE Main Students:
- Time Required: 12-15 days (2 hours/day)
- Master calorimetry problems first (highest weightage)
- Practice 100+ numerical on mixing and phase change
- Learn all standard values (c, L_f, L_v, k for common materials)
- Focus on Newton's cooling average rate formula
- Thermal expansion - focus on railway track, pendulum problems
- Heat conduction - series/parallel combination is key
For JEE Advanced Students:
- Time Required: 18-20 days (3 hours/day)
- Deep dive into multi-step calorimetry (ice→steam complete)
- Master complex compound bar problems (3+ rods, triangle arrangement)
- Practice Newton's cooling with exponential form
- Combine radiation with other heat transfer modes
- Study thermal stress and real-life applications
- Solve PYQs from last 10 years (pattern recognition)
⚠️ Top 10 Mistakes to Avoid in JEE Exam
-
❌
Not converting units: Always convert to SI (kg, K, J) before calculation
-
❌
Celsius in formulas: Use Kelvin in Stefan's law, gas equations
-
❌
Forgetting phase change: Heat = sensible + latent (both needed!)
-
❌
Wrong β, γ relation: Remember β=2α, γ=3α (not 2, 3 directly)
-
❌
Mixing sign errors: Heat lost is negative, heat gained is positive
-
❌
Newton's law for large ΔT: Valid only when ΔT is small (<30°C)
-
❌
Compound bar confusion: Series → same Q, different T. Parallel → same ΔT, different Q
-
❌
Emissivity in Stefan's law: Don't forget 'e' factor for gray bodies
-
❌
Average temperature: Use (T₁+T₂)/2 in Newton's cooling rate formula
-
❌
Thermal stress: Remember to use ΔL prevented = αL₀ΔT, then F = YAΔl/l
🔗 How This Chapter Connects with Other Topics
With Thermodynamics:
First law connects heat and work. Thermal properties provides foundation for understanding heat engines, refrigerators, and entropy changes.
With Kinetic Theory:
Specific heat capacities (C_v, C_p) are explained using kinetic theory. Temperature is linked to molecular kinetic energy.
With Modern Physics:
Black body radiation leads to quantum theory (Planck's law), photoelectric effect. Wien's law connects to quantum nature of light.
With Waves:
Thermal radiation is electromagnetic waves. Wavelength calculations use wave concepts.
⚡ Last Minute Revision Points (Day Before Exam)
Temperature & Heat
- • °C = K - 273
- • °F = (9/5)°C + 32
- • Heat ≠ Temperature
- • 1 cal = 4.186 J
- • Water at 4°C → max density
Expansion & Calorimetry
- • β = 2α, γ = 3α
- • Q = mcΔT (no phase change)
- • Q = mL (phase change)
- • Heat lost = Heat gained
- • c_water highest (4200)
Heat Transfer
- • Conduction: Q/t = kAΔT/L
- • Radiation: P = σAT⁴
- • Newton: dT/dt ∝ (T-T₀)
- • Wien: λ_m T = const
- • Good absorber = Good emitter