Thermodynamics - Complete Guide for JEE 2025-26
Why Thermodynamics is Most Important for JEE?
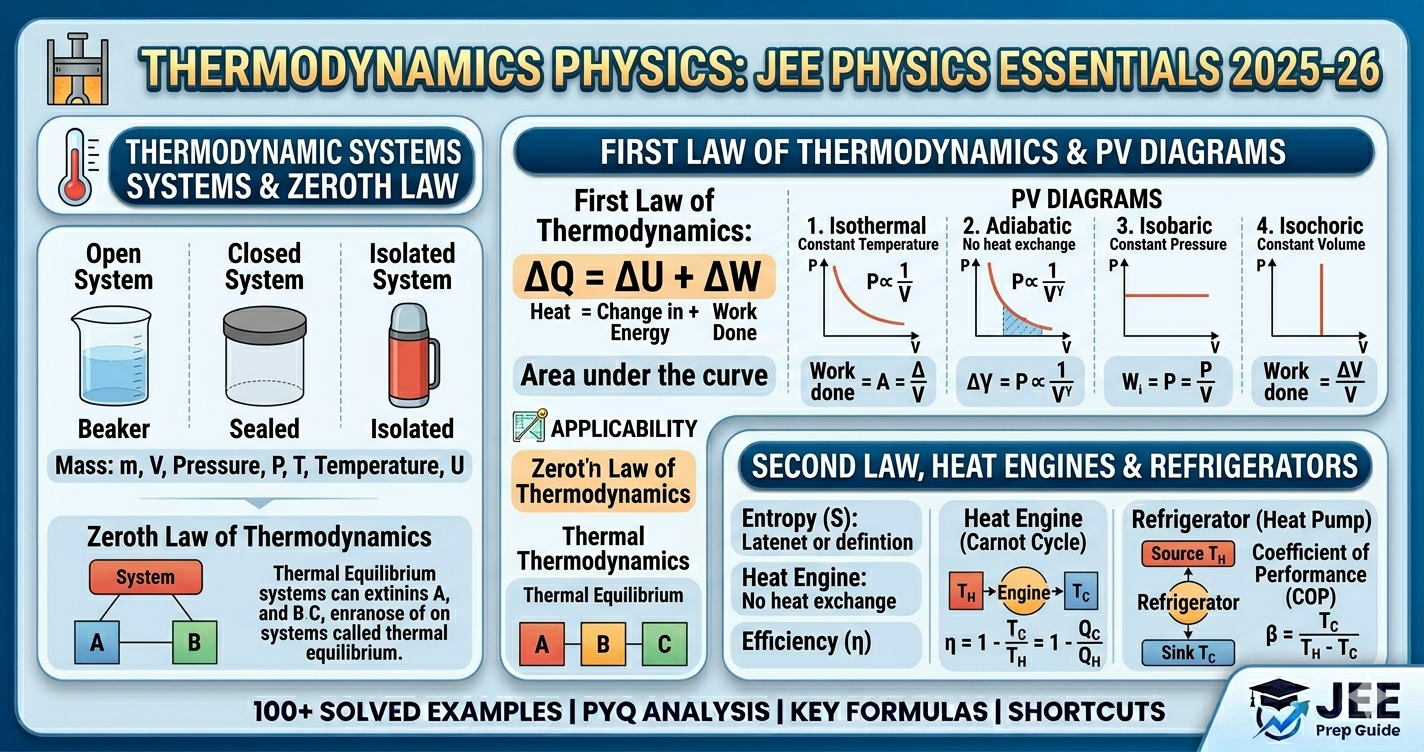
Thermodynamics is one of the highest-scoring chapters in JEE Physics with 8-12% weightage. It's conceptually straightforward once you understand the laws and processes.
- First Law - Foundation for 60% of problems (Q = ΔU + W)
- Thermodynamic Processes - Know all 4 types thoroughly
- PV Diagrams - Work = Area under curve (visual problems)
- Heat Engines - Efficiency calculations (guaranteed questions)
- Carnot Engine - Maximum efficiency benchmark
- Cp, Cv, γ - Essential for adiabatic process problems
In JEE Advanced, expect complex cyclic processes and combined problems with kinetic theory.
Master These Formulas for 100% Accuracy
1. The Golden Equation
Q = ΔU + W (First Law)
ΔU = nCvΔT for ALL processes of ideal gas
2. Process Equations
- • Isothermal: PV = const, W = nRT ln(V₂/V₁)
- • Adiabatic: PVᵞ = const, W = (P₁V₁-P₂V₂)/(γ-1)
- • Isobaric: W = PΔV = nRΔT
- • Isochoric: W = 0
3. Heat Engine Efficiency
η = 1 - Q₂/Q₁ = 1 - T₂/T₁ (Carnot)
4. Mayer's Relation
Cp - Cv = R
📚 How to Master Thermodynamics for JEE?
For JEE Main Students:
- Time Required: 15-18 days (3 hours/day)
- Master the First Law and sign conventions first
- Practice 100+ problems on work done in different processes
- Memorize γ values: 5/3 (mono), 7/5 (dia), 4/3 (poly)
- Focus on Carnot engine efficiency problems
- Learn to read PV diagrams and calculate area
- Solve all NCERT examples and exercises
For JEE Advanced Students:
- Time Required: 20-25 days (4 hours/day)
- Deep dive into polytropic processes (PVⁿ = const)
- Master complex cyclic processes with multiple stages
- Practice deriving efficiency for custom cycles
- Study entropy and Second Law applications
- Solve combined problems (thermo + kinetic theory)
- Practice integer-type and matrix-match questions
- Solve last 15 years JEE Advanced PYQs
⚠️ Top 10 Mistakes to Avoid in JEE Exam
-
❌
Sign convention confusion: W positive for expansion (work BY system), Q positive for heat absorbed
-
❌
Using ΔU = nCpΔT: Wrong! ΔU = nCvΔT for ALL processes
-
❌
Temperature in Celsius: Always use Kelvin in thermodynamics formulas
-
❌
Forgetting Q = 0 in adiabatic: So W = -ΔU (work done at expense of internal energy)
-
❌
Confusing η and COP: η < 1 always, but COP can be > 1
-
❌
Wrong γ value: Air is diatomic (γ = 1.4), not monoatomic
-
❌
Cyclic process ΔU: ΔU = 0 for complete cycle, so Q_net = W_net
-
❌
Carnot efficiency confusion: η = 1 - T₂/T₁ (not T₂/T₁)
-
❌
Work in cyclic process: W = Area enclosed (clockwise +, anticlockwise -)
-
❌
Free expansion: Q = W = 0, ΔU = 0, ΔT = 0 (not adiabatic compression!)
🔗 How Thermodynamics Connects with Other Topics
With Kinetic Theory of Gases:
Internal energy U = (f/2)nRT comes from kinetic theory. Cp, Cv values derived from degrees of freedom. Many JEE Advanced questions combine both chapters.
With Thermal Properties of Matter:
Specific heat, latent heat from thermal properties are used in thermodynamics. Calorimetry problems often use First Law concepts.
With Chemistry (Physical Chemistry):
Enthalpy (H = U + PV), Gibbs free energy, Hess's law - all based on thermodynamics. Sign conventions differ - be careful!
With Mechanical Properties:
Bulk modulus appears in sound velocity formula with γ. Adiabatic bulk modulus B_ad = γP.
⚡ Last Minute Revision Points (Night Before Exam)
Laws & Definitions
- • Zeroth Law: Thermal equilibrium
- • First Law: Q = ΔU + W
- • Second Law: η < 1 always
- • Internal energy is state function
- • Q, W are path functions
Key Numbers to Remember
- • R = 8.314 J/mol·K
- • γ_mono = 5/3 = 1.67
- • γ_dia = 7/5 = 1.4
- • ln(2) = 0.693
- • ln(10) = 2.303
Quick Checks
- • Isothermal: ΔU = 0, Q = W
- • Adiabatic: Q = 0, W = -ΔU
- • Isochoric: W = 0, Q = ΔU
- • Isobaric: All three non-zero
- • Cyclic: ΔU = 0, Q = W
🧠 Complete Chapter Mind Map
Zeroth Law
Thermal Equilibrium
Temperature Definition
First Law
Q = ΔU + W
Energy Conservation
Processes
Isothermal, Adiabatic
Isobaric, Isochoric
Second Law
Heat Engines
Entropy, Carnot
Cp - Cv = R
η = 1 - T₂/T₁
ΔS ≥ 0